乳糜泻(专业版)
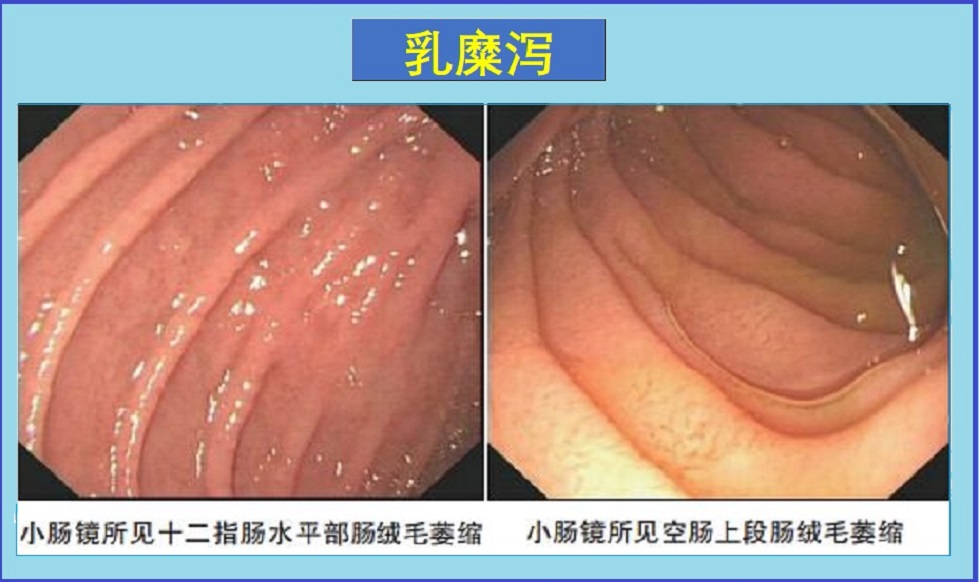
乳糜泻,也称为麦胶性肠病,是发生在消化道的一种自体免疫性疾病。在乳糜泻患者中,吃含麸质的食物会损害小肠吸收营养的绒毛。
其他名称:腹腔疾病;麦胶性肠病;麸质敏感性肠病
英文名称:Celiac Disease,Celiac Sprue,Gluten-Sensitive Enteropathy
乳糜泻的确切病因尚不清楚。分析认为,其发病原因存在下列因素:
其他可增加乳糜泻发生的危险因素,包括如下:
调整饮食与生活方式
营养与草本综合干预
以下是基于循证医学和循证营养学有关文献综合的结果。
有助于防控乳糜泻的营养和草本补充剂,主要包括如下:
1.维生素D:
维生素D缺乏是乳糜泻的常见问题,与一系列肌肉骨骼疾病有关,包括骨痛、肌肉疾病(肌病)、骨密度损失、骨质减少和骨质疏松症1-3。一项研究发现,患有乳糜泻的64%的男性和71%的女性维生素D水平较低4。然而,重要的是,这项研究将维生素D的“缺乏”定义为低于10ng/mL的25-羟基维生素D,这远低于被认为是最佳的水平。此外,有证据表明,乳糜泻患者的血清维生素D水平随着年龄的增长而降低,即使在阳光明媚的气候下也会出现低水平5。一份病例报告描述了一名患有肌病且维生素D水平低于4ng/dL的乳糜泻患者。患者开始无麸质饮食,并开始补充维生素D,剂量为30万IU每两周一次。患者的症状在一周内得到缓解3。
补充维生素D和钙,以及无麸质饮食,已被证明可以显著改善乳糜泻患者的骨骼状况6,7,2,并且广泛建议乳糜泻病人补充维生素D或钙8,9,2。维生素D除了在肌肉骨骼系统中的作用外,还有许多其他功能,特别是调节免疫系统和抗炎作用。来自实验室研究的数据为维生素D在减少人类全身炎症和预防自身免疫性疾病方面的潜力提供了证据10-12。具体而言,维生素D抑制促炎细胞因子(如干扰素γ和TNF-α)的分泌,同时促进抗炎细胞因子(包括IL-4、IL-5和IL-10)的产生13。
25-羟基维生素D的血液测试可以确定基线水平,并监测补充维生素D和无麸质饮食引起的水平变化。
2.钙:
在乳糜泻中,肠道内壁的损伤通常会导致钙吸收不良和血清钙水平下降2,14,15,8,然后导致骨质流失、骨质减少、骨质疏松和骨折风险增加16,17。在无麸质饮食中,钙水平和骨密度会改善,但只有在一到两年后才会提高18,19。然而,无麸质饮食可能钙含量低,并可能导致缺钙20-22和随后的低骨密度。因此,补充钙对乳糜泻患者很重要2,8,9,23。
3.铁:
缺铁性贫血是乳糜泻最常见的肠外症状之一18,24;也可能出现缺铁性贫血。尽管缺铁通常可以通过无麸质饮食来纠正,但在小肠内壁愈合后,铁水平的恢复可能需要6-12个月18。缺铁性血液测试,包括全血细胞计数和铁蛋白水平,在诊断中很重要25,补铁被认为是乳糜泻患者必要护理的一部分,部分原因是无麸质饮食可能导致营养不足20,26,27。
4.维生素B族:
缺乏叶酸和维生素B12和维生素B6在乳糜泻中很常见28-30,18,补充这些B族维生素以及烟酸(维生素B3)和维生素B2(核黄素),被认为是乳糜泻患者常规护理的重要组成部分31,32,27。在一项研究中,叶酸和5’-磷酸吡哆醛(维生素B6的活性形式)分别在37%和20%的成年乳糜泻患者中缺乏,这些患者已经无麸质饮食10年,并且活组织检查证明他们已经康复。在这项研究的参与者中,乳糜泻患者的平均每日叶酸和维生素B12摄入量显著低于对照组33。
同型半胱氨酸是一种氨基酸衍生物,可损伤血管内壁并促进动脉粥样硬化疾病34,35。同型半胱氨酸升高通常归因于缺乏维生素B12、B6和叶酸,而这些是同型半胱氨酸代谢所必需的36,37。几项研究表明,乳糜泻患者血液中同型半胱氨酸水平升高38,39。一项研究表明,与不使用维生素补充剂的患者相比,服用维生素补充剂的乳糜泻患者血液中叶酸、维生素B6和B12水平较高,同型半胱氨酸水平较低31。叶酸和维生素B12缺乏会导致维生素B缺乏性贫血,这在乳糜泻中很常见,高达34%的未经治疗的患者会出现这种情况40,41。
5.镁:
镁是人体内数百种酶促反应的必需矿物质42。镁也是维生素D和钙的适当代谢所必需的,因此也是骨骼健康所必需的43,44。
镁缺乏在乳糜泻中的重要性早在上世纪60年代首次被认识到,当时一份详细的病例报告显示,一名年轻成年乳糜泻患者在开始无麸质饮食后,镁的吸收和状态显著改善45。另一项研究调查了患有乳糜泻的儿童和青少年的镁状况。虽然所有患有典型乳糜泻(有吸收不良)的患者都缺乏镁,但只有五分之一的无麸质饮食患者(没有吸收不良)和五分之一患有无症状乳糜泻患者(没有吸收不良)的镁含量低46。后来的一项研究检测了41名患有乳糜泻或正常肠绒毛的儿童和青少年的镁,这些儿童和青少年平均11年无麸质饮食;28名未经治疗的非典型乳糜泻患者和8名对照组也被纳入研究。研究人员发现,通过红细胞镁水平评估,14.6%的无麸质饮食患者和25%的未经治疗的患者缺乏镁47。
一项对23名成年乳糜泻患者的早期研究发现,尽管他们没有症状(没有吸收不良),而且都在无麸质饮食中,但他们的细胞内镁水平都有所下降。这些人的镁治疗导致红细胞镁和骨密度增加,这表明镁缺乏可能在乳糜泻患者骨质疏松症的发展中发挥作用48。筛查镁的状况和缺乏,以及补充镁和增加饮食,被认为是乳糜泻患者护理的重要方面18,25,27。
6.锌:
锌是一种重要的营养矿物质,在体内300多种不同的酶促反应中起催化作用49。锌缺乏在未经治疗的乳糜泻中很常见,而且这种缺乏并不总是通过无麸质饮食来纠正;一些研究认为补锌是乳糜泻治疗的必要组成部分18,27,20。缺锌可能是乳糜泻生长、免疫功能、伤口愈合不良和皮肤问题的罪魁祸首。
一项对未经治疗和治疗的乳糜泻患者的研究综述发现,通过血浆或血清锌测量,锌缺乏的患病率显著。11项针对未经治疗的患有乳糜泻的成人和儿童的独立研究发现,高达100%的患者缺锌。9项针对乳糜泻患者的无麸质饮食研究中,有三项长达三个月至十年,发现20-40%的患者持续缺锌。仅在无麸质饮食中,缺锌通常需要一整年的时间才能解决,有人建议监测乳糜泻患者的血清锌水平18,32,50。
7.维生素E:
维生素E是一种重要的脂溶性维生素和自由基清除剂,在保护细胞膜免受氧化损伤方面发挥重要作用51。几项研究报告称,乳糜泻患者的维生素E水平较低50,52,53。维生素E缺乏也与乳糜泻神经症状的发展有关53-55。补充维生素E和无麸质饮食已被证明可以改善乳糜泻中维生素E缺乏引起的神经损伤53-55。
8.维生素A:
维生素A是一种脂溶性营养素,乳糜泻患者可能因吸收不良而缺乏维生素A。维生素A对正常免疫功能、视力和基因表达很重要56。乳糜泻维生素A缺乏症的治疗方法是无麸质饮食和补充27。
一项针对新诊断的乳糜泻患者的研究发现,其中7.5%的患者缺乏维生素A,而健康对照组则没有40。一项案例研究重申了维生素A的重要性,即使在控制性乳糜泻病例中也是如此。一名64岁的男性,经活检证实腹腔疾病在无麸质饮食中得到控制,最近出现腹泻,一只眼睛发红和模糊。在用药物治疗他的眼部症状的三周里,他的病情恶化了。维生素A的测试显示出严重缺乏。患者接受了含有100 ,000IU的维生素A,并且在一周内观察到视力和眼睛状况的显著改善。该患者的随访包括定期维生素A治疗57。
9.维生素K:
维生素K是脂溶性维生素之一,在乳糜泻中会因吸收不良而缺乏27,58,59。维生素K缺乏会导致容易出现瘀伤,这是乳糜泻的一种非典型症状;维生素K缺乏也已被证明会增加乳糜泻患者出现血栓和出血事件的风险60-63。在2012年一项针对患有乳糜泻的儿童和青少年的研究中,对维生素K的饮食摄入量进行了评估。在诊断时,41%的患者的摄入量低于推荐量的一半,而在无麸质饮食一年后,31%的患者的摄入仍然不足58。
已发现患有症状性和无症状性乳糜泻的儿童在诊断时骨量减少58,64,65。维生素K增强钙代谢,因此在骨骼健康中发挥重要作用66,67。由于钙、维生素K和D的饮食摄入通常不足,包括无麸质饮食的患者,因此建议在诊断为乳糜泻时常规补充维生素K58。
10.硒:
硒是谷胱甘肽过氧化物酶的组成部分,谷胱甘肽过氧化物酶在细胞抗氧化防御中发挥重要作用68。在一项针对30名患有乳糜泻的儿童的研究中,80%的儿童血清硒水平低于正常范围69。硒对甲状腺激素调节至关重要,乳糜泻中的硒缺乏与自身免疫性甲状腺疾病的风险增加有关70,71。
11.益生菌:
益生菌是定居在肠道中的有益微生物,在肠道健康中发挥着重要作用;在患有乳糜泻的患者中,这些生物体的平衡似乎紊乱了72,73。在一项为期三周的随机对照试验中,在未经治疗的乳糜泻患者中补充婴儿双歧杆菌NLS超级菌株可改善消化不良、便秘和胃酸倒流74。
目前正在开发各种益生菌菌株,专门针对乳糜泻的免疫和肠道损伤机制。乳双歧杆菌菌株已被证明可以保护培养的肠道上皮细胞免受面筋诱导的损伤,可能是通过阻断面筋诱导的肠道通透性增加75。此外,在一项实验室研究中,八种不同益生菌菌株的混合物(ACTIAL益生菌专有品牌VSL#3),包括几种乳酸菌和双歧杆菌,被证明可以广泛预消化小麦粉中的面筋蛋白。当乳糜泻患者的肠道活检样本暴露于这些预先消化的面筋蛋白时,免疫细胞的募集减少。研究人员得出结论,益生菌配方降低或消除了小麦面筋的毒性76。
12.维生素C:
维生素C是一种水溶性自由基清除剂,对全身组织的结构完整性也至关重要77。在一项实验中,用添加和不添加维生素C的麸质醇溶蛋白组分对乳糜泻患者的培养肠活检进行攻击。向培养细胞中添加维生素C降低了促炎细胞因子TNF-α、IL-6和干扰素γ的分泌。维生素C还完全抑制促炎细胞因子IL-15的分泌,IL-15与乳糜泻中发生的损伤密切相关78,79。
13.欧米伽3脂肪酸:
ω-3脂肪酸具有重要的生物抗炎作用80,81。当通过食物或补充剂摄入时,ω-3脂肪酸与更具促炎性的ω-6脂肪酸花生四烯酸竞争,以结合到富含脂质的细胞膜中82-84。
一项对新诊断为乳糜泻的成年人血清脂肪酸组成的研究发现,ω-3脂肪酸的浓度明显低于对照组。在无麸质饮食一年并获得临床缓解后,ω-3脂肪酸水平增加,但仍远低于对照值85。一项针对患有乳糜泻和1型糖尿病的儿童的研究发现,他们的ω-3脂肪酸水平(包括EPA和DHA)显著低于对照组86。
一项使用肠上皮细胞培养物的研究支持口服补充DHA在减少乳糜泻肠道炎症方面的潜在益处。作为对麸质引发的炎症的反应,肠细胞释放花生四烯酸。而DHA被证明可以阻断花生四烯酸的释放以及由此产生的炎症级联反应,后者促进疾病的发展87,88。ω-3脂肪酸减少炎症的另一种机制是通过抑制核因子κB(NF-ĸB),一种促炎介质89。DHA还刺激一种名为PPARγ的细胞受体,该受体激活减少促炎细胞因子产生的基因90。
14.L肉碱:
L-肉碱是一种在人体内由赖氨酸和蛋氨酸制成的化合物,在饮食中也主要从肉类、鱼类和乳制品等动物性食品中获得91。在一项随机对照试验中,连续六个月每天补充2g L-肉碱可显著改善患有乳糜泻的成年人的疲劳。L-肉碱的剂量是安全且耐受性良好的92)。
L-肉碱需要将长链脂肪酸穿梭到线粒体(细胞“能量站”)中,以产生肌肉能量。乳糜泻的肠道损伤导致吸收表面积减少,增加了L-肉碱缺乏的风险。在一项研究中,未经治疗的乳糜泻患者的血清L-肉碱浓度较低,在无麸质饮食后恢复正常93。另一项研究发现患有乳糜泻的儿童血清肉碱水平较低69。L-肉碱缺乏可能会导致疲劳、虚弱和体重增加困难,这些都是乳糜泻的常见症状93,94。
15.谷胱甘肽:
谷胱甘肽是人体对抗破坏性自由基最强大的防御系统之一,并对肝脏的解毒功能至关重要95,96。几项研究报告称,与对照组相比,乳糜泻患者肠道内壁97,98和外周血98,99中的谷胱甘肽浓度显著降低。结果,肠内膜中脂质过氧化物或酸腐脂肪的浓度增加。脂质过氧化物可促进组织中的一系列氧化损伤,导致肠道中的组织损伤,这是乳糜泻的特征,并增加乳糜泻患者患癌症的风险97-99。
通过直接补充谷胱甘肽100或使用生理前体如半胱氨酸101,102和N-乙酰半胱氨酸95,96,以及α-硫辛酸、硒和乳清蛋白103-105,可以增加人体的谷胱甘肽储备。
16.消化酶(胰酶):
胰腺外分泌功能不全是一种没有足够的消化酶分泌到小肠中的情况106,在对无麸质饮食没有反应的乳糜泻患者中相对常见25。小肠受损可能会抑制胰腺分泌消化酶的能力。一项研究发现,即使在无麸质饮食后腹泻仍持续的病例中,胰腺功能不全的实验室症状发生率明显更高。接受胰酶治疗的患者症状减轻了75%。另一项研究发现,小肠绒毛萎缩的严重程度与胰腺外分泌功能不全的实验室体征显著相关。在一项研究中,超过一半的乳糜泻患者出现胰腺酶功能不全,而在无麸质饮食一年后恢复正常107-109。
17.其他支持:
以下干预措施尚未在乳糜泻的背景下进行彻底研究,但可能有助于支持肠道健康或对抗炎症反应或细胞代谢紊乱,这被认为是乳糜泻几种表现的病理基础。
更多可点击其个性化综合干预方案如下:
以及参阅本网如下专文的相关内容:
医疗干预
治疗乳糜泻需要面临如下问题:
如果孩子患腹腔疾病的风险增加,日常生活中要尽量食用无麸质食品。
越早吃无麸质饮食,对肠道的损伤就越小。
参考文献:
1. Mulder CJ et al. Celiac disease presenting as severe osteopenia. Hawaii medical journal. Nov 2011;70(11):242-244.
2. Rastogi A et al. Celiac disease: a missed cause of metabolic bone disease. Indian J Endocrinol Metab. 2012;Sep-Oct;16(5):780-785.
3. Karaahmet OZ et al. Myopathy related to vitamin D deficiency in patient with celiac disease. Muscle & nerve. Jul 2014;50(1):147-148.
4. Kemppainen T et al. Osteoporosis in adult patients with celiac disease. Bone. 1999;24(3):249-255.
5. Lerner A et al. The clinical significance of 25OH-vitamin D status in celiac disease. Clinic Rev Allerg Immunol. 2012;42:322-330.
6. Duerksen DR. Dramatic effect of vitamin D supplementation and a gluten-free diet on bone mineral density in a patient with celiac disease. J Clin Densitom. 2012;Jan-Mar;15(1):120-123.
7. Bianchi ML, Bardella MT. Bone in celiac disease. Osteoporos Int. 2008;19:1705-1716.
8. Szymczak J et al. Low bone mineral density in adult coeliac disease. Endokrynol Pol. 2012;63(4):270-6.
9. Lucendo AJ et al. Bone mineral density in adult coeliac disease: an updated review. Rev Esp Enferm Dig. 2013;Mar;105(3):154-62.
10. Richards JB et al. Higher serum vitamin D concentrations are associated with longer leukocyte telomere length in women. The American journal of clinical nutrition. Nov 2007;86(5):1420-1425.
11. Kriegel MA et al. Does vitamin D affect risk of developing autoimmune disease?: a systematic review. Semin Arthritis Rheum. 2011;40:512-531.
12. Agmon-Levin N et al. Vitamin D in systemic and organ-specific autoimmune diseases. Clinic Rev Allerg Immunol. 2013;45:256-266.
13. Prietl B et al. Vitamin D and immune function. Nutrients. 2013;5:2502-2521.
14. Pantaleoni S et al. Bone mineral density at diagnosis of celiac disease and after 1 year of gluten-free diet. Scientific World Journal. 2014;2014:173082.
15. Babar MI et al. Celiac disease and celiac crisis in children. J Coll Physicians Surg Pak. 2011;Aug;21(8):487-90.
16. AGA. American Gastroenterological medical position statement: celiac sprue. Gastroenterology. 2001;120(6):1522-5.
17. Pantaleoni S et al. Bone mineral density at diagnosis of celiac disease and after 1 year of gluten-free diet. Scientific World Journal. 2014;2014:173082.
18. Caruso R et al. Appropriate nutrient supplementation in celiac disease. Ann Med. 2013;Dec;45(8):522-31.
19. Molteni N et al. Intestinal calcium absorption as shown by stable strontium test in celiac disease before and after gluten-free diet. Am J Gastroenterol. 1995; Nov;90(11):2025-8.
20. Kupper C. Dietary guidelines and implementation for celiac disease. Gastroenterology. Apr 2005;128(4 Suppl 1):S121-127.
21. UCMC. The University of Chicago Celiac Disease Center. Jump start your gluten-free diet. Available at: http://www.cureceliacdisease.org/living-with-celiac/resources/jump-start-your-gluten-free-diet-ebook. Updated 2013. Accessed 10/17/2014.
22. Blazina S et al. Bone mineral density and importance of strict gluten-free diet in children and adolescents with celiac disease. Bone. 2010;Sep;47(3):598-603.
23. Mancini LA et al. Celiac disease and the athlete. Curr Sports Med Rep. 2011;Mar-Apr;10(2):105-8.
24. Horowitz S. Celiac Disease: New Directions in Diagnosis, Treatment, and Prevention. Alternative and Complementary Therapies. 2011;17(2):92-98.
25. Ferri FF. Ferri's Clinical Advisor. Celiac Disease. Available at: www.clinicalkey.com. Copyright 2015. Accessed 2/3/2015.
26. Thompson T. Folate, iron, and dietary fiber contents of the gluten-free diet. Journal of the American Dietetic Association. Nov 2000;100(11):1389-1396.
27. Lidums I et al. First Consult. Celiac disease. Available at: www.clinicalkey.com. Copyright 2015 by Elsevier Inc.
28. Dickey W. Low serum vitamin B12 is common in coeliac disease and is not due to autoimmune gastritis. Eur J Gastroenterol Hepatol. 2002;14(4):425-427.
29. Haapalahti M et al. Nutritional status in adolescents and young adults with screen-detected celiac disease. J Pediatr Gastroenterol of Nutr. 2005;May;40:566-570.
30. Tikkakoski S et al. Undiagnosed coeliac disease and nutritional deficiencies in adults screened in primary health care. Scand J Gastroenterol. 2007;Jan;’42(1):60-65.
31. Hadithi M et al. Effect of B vitamin supplementation on plasma homocysteine levels in celiac disease. World J Gastroenterol. 2009;Feb 28;15(8):955-960.
32. Wierdsma NJ, van Bokhorst-de van der Schueren MA, Berkenpas M, et al. Vitamin and mineral deficiencies are highly prevalent in newly diagnosed celiac disease patients. Nutrients. 2013;Sep 30;5(10):3975-92.
33. Hallert C et al. Evidence of poor vitamin status in coeliac patients on a gluten-free diet for 10 years. Aliment Pharmacol Ther. 2002;16:1333-1339.
34. Eren E et al. Homocysteine, Paraoxonase-1 and Vascular Endothelial Dysfunction: Omnibus viis Romam Pervenitur. Journal of clinical and diagnostic research: JCDR. Sep 2014;8(9):Ce01-04.
35. Pushpakumar S et al. Endothelial dysfunction: the link between homocysteine and hydrogen sulfide. Current medicinal chemistry. 2014;21(32):3662-3672.
36. Higdon J. Linus Pauling Institute. Micronutrient Information Center. Vitamin B12. Available at: http://lpi.oregonstate.edu/infocenter/vitamins/vitaminB12/. Last updated 1/2014a. Accessed 3/5/2015.
37. Higdon J. Linus Pauling Institute. Micronutrient Information Center. Vitamin B6. Available at: http://lpi.oregonstate.edu/infocenter/vitamins/vitaminB6/. Last updated 5/2014b. Accessed 3/5/2015.
38. Hallert C et al. Evidence of poor vitamin status in coeliac patients on a gluten-free diet for 10 years. Aliment Pharmacol Ther. 2002;16:1333-1339.
39. Dickey W et al. Homocysteine and related B-vitamin status in coeliac disease: effects of gluten exclusion and histological recovery. Scand J Gastroenterol. 2008;43:682-688.
40. Wierdsma NJ et al. Vitamin and mineral deficiencies are highly prevalent in newly diagnosed celiac disease patients. Nutrients. 2013;Sep 30;5(10):3975-92.
41. Halfdanarson TR et al. Hematologic manifestations of celiac disease. Blood. 2007 Jan 15;109(2):412-421.
42. Higdon J. Linus Pauling Institute. Micronutrient Information Center. Micronutrient Information Center. Magnesium. Available at: http://lpi.oregonstate.edu/infocenter/minerals/magnesium/. Last updated 10/2013a. Accessed 2/11/2015.
43. Zofkova I et al. The relationship between magnesium and calciotropic hormones. Magnes Res. 1995;Mar;8(1):77-84.
44. Hardwick LL et al. Magnesium absorption: mechanisms and the influence of vitamin D, calcium and phosphate. The Journal of nutrition. Jan 1991;121(1):13-23.
45. Goldman AS et al. Magnesium deficiency in celiac disease. Pediatrics. Jun 1962;29:948-952.
46. Rujner J et al. Magnesium status in children and adolescents with celiac disease. Wiad Lek. 2001;54(5-6):277-285.
47. Rujner J et al. Magnesium status in children and adolescents with coeliac disease without malabsorption symptoms. Clinical nutrition (Edinburgh, Scotland). Oct 2004;23(5):1074-1079.
48. Rude RK et al. Magnesium deficiency: possible role in osteoporosis associated with gluten-sensitive enteropathy. Osteoporos Int. 1996;6(6):453-61.
49. Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Zinc. Available at: http://lpi.oregonstate.edu/infocenter/minerals/zinc/. Last updated 6/2013b. Accessed 3/5/2015.
50. Guevara Pacheco G et al. [Micronutrient deficiencies and celiac disease in Pediatrics]. Arch Argent Pediatr. 2014;Oct;112(5):457-63.
51. Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Vitamin E. Available at: http://lpi.oregonstate.edu/infocenter/vitamins/vitaminE/. Last updated 6/2008. Accessed 3/5/2015.
52. Odetti P et al. Oxidative stress in subjects affected by celiac disease. Free Radic Res. 1998;Jul;29(1):17-24.
53. Henri-Bhargava A et al. Neurologic impairment due to vitamin E and copper deficiencies in celiac disease. Neurology. 2008;Sep 9;71(11):860-1.
54. Kleopa KA et al. Reversible inflammatory and vacuolar myopathy with vitamin E deficiency in celiac disease. Muscle Nerve. 2005;31:260-265.
55. Mauro A et al. Cerebellar syndrome in adult celiac disease with vitamin E deficiency. Acta Neurol Scand. 1991;84:167-170.
56. Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Vitamin A. Available at: http://lpi.oregonstate.edu/infocenter/vitamins/vitaminA/. Last updated 11/2007a. Accessed 3/5/2015.
57. Alwitry A. Vitamin A deficiency in coeliac disease. The British Journal of Ophthalmology. 2000;84(9):1075-1075.
58. Mager DR et al. Vitamin D and K status influences bone mineral density and bone accrual in children and adolescents with celiac disease. Eur J Clin Nutr. 2012;66:488-495.
59. Kelly CP et al. ePocrates online. Celiac disease. Available at: https://online.epocrates.com/noFrame/showPage?method=diseases&MonographId=636. Last updated 4/11/2014. Accessed 2/3/2015.
60. Berthoux E et al. [Adult celiac disease with thrombosis: a case series of seven patients. Role of thrombophilic factors]. La Revue de medecine interne / fondee ... par la Societe nationale francaise de medecine interne. Oct 2011;32(10):600-604.
61. Chen CS et al. Coagulopathy due to celiac disease presenting as intramuscular hemorrhage. Journal of general internal medicine. Nov 2007;22(11):1608-1612.
62. Vaynshtein G et al. Celiac sprue presenting as severe hemorrhagic diathesis due to vitamin K deficiency. The Israel Medical Association journal : IMAJ. Dec 2004;6(12):781-783.
63. Lerner A et al. Hypercoagulability in celiac disease--an update. Autoimmunity reviews. Nov 2014;13(11):1138-1141.
64. Turner J et al. Prevalence of metabolic bone disease in children with celiac disease is independent of symptoms at diagnosis. J Pediatr Gastroenterol Nutr. 2009;49:589-593.
65. Rajani S et al. The changing frequency of celiac disease diagnosed at the Stollery Children’s Hospital. Can J Gastroenterol. 2010;24(2):109-112.
66. Zittermann A. Effects of vitamin K on calcium and bone metabolism. Current opinion in clinical nutrition and metabolic care. Nov 2001;4(6):483-487.
67. Booth SL. Roles for vitamin K beyond coagulation. Annu Rev Nutr. 2009;29:89-110.
68. Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Selenium. Available at: http://lpi.oregonstate.edu/infocenter/minerals/selenium/. Last updated 11/2007b. Accessed 3/5/2015.
69. Yuce A et al. Serum carnitine and selenium levels in children with celiac disease. Indian J Gastroenterol. 2004;May-June;23:87-88.
70. Stazi AV et al. Selenium deficiency in celiac disease: risk of autoimmune thyroid diseases. Minerva Med. 2008;Dec;99(6):643-53.
71. Stazi AV et al. Selenium status and over-expression of interleukin-15 in celiac disease and autoimmune thyroid diseases. Ann Ist Super Sanita. 2010;46(4):389-399.
72. Pozo-Rubio T et al. Immune development and intestinal microbiota in celiac disease. Clin Dev Immunol. 2012;2012:654143.
73. Round JL et al. The gut microbiota shapes intestinal immune responses during health and disease. Nat Rev Immunol. 2009;May;9:313-323.
74. Smecuol E et al. Exploratory, randomized, double-blind, placebo-controlled study on the effects of Bifidobacterium infantis natren life start strain super strain in active celiac disease. J Clin Gastroenterol. 2013;47:139-147.
75. Lindfors K et al. Live probiotic Bifidobacterium lactis bacteria inhibit the toxic effects induced by wheat gliadin in epithelial cell culture. Clin Exp Immunol. 2008;152:552-558.
76. De Angelis M et al. VSL#3 probiotic preparation has the capacity to hydrolyze gliadin polypeptides responsible for Celiac Sprue. Biochim Biophys Acta. 2006;Jan;1762(1):80-93.
77. Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Vitamin C. Available at: http://lpi.oregonstate.edu/infocenter/vitamins/vitaminC/#function. Last updated 11/2013c. Accessed 3/5/2015.
78. Bernardo D et al. Ascorbate-dependent decrease of the mucosal immune inflammatory response to gliadin in celiac disease patients. Allergol Immunopathol (Madr). 2012;40(1):3-8.
79. Gujral N et al. Celiac disease: Prevalence, diagnosis, pathogenesis and treatment. World J Gastroenterol. 2012;18(42):6036-6059.
80. Skulas-Ray AC. Omega-3 fatty acids and inflammation: A perspective on the challenges of evaluating efficacy in clinical research. Prostaglandins & other lipid mediators. 2015 Jan-Mar:116-117:104-11.
81. Calder PC. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochimica et biophysica acta. Apr 2015;1851(4):469-484.
82. Westphal C et al. CYP-eicosanoids – a new link between omega-3 fatty acids and cardiac disease? Prostaglandins Other Lipid Mediat. 2011;96:99-108.
83. Calder PC. Immunomodulation by omega-3 fatty acids. Prostaglandins Leukot Essent Fatty Acids. 2007;77:327-335.
84. Surette ME. The science behind dietary omega-3 fatty acids. Canadian Medical Association journal. Jan 15 2008;178(2):177-180.
85. Solakivi T et al. Serum fatty acid profile in celiac disease patients before and after a gluten-free diet. Scand J Gastrolenterol. 2009;44:826-830.
86. Tarnok A et al. Low n-3 Long-Chain Polyunsaturated Fatty Acids in Newly Diagnosed Celiac Disease in Children With Preexisting Type 1 Diabetes Mellitus. Journal of pediatric gastroenterology and nutrition. Feb 2015;60(2):255-258.
87. Vincentini O et al. Docosahexaenoic acid modulates in vitro the inflammation of celiac disease in intestinal epithelial cells via the inhibition of cPLA2. Clin Nutr. 2011;30:541-546.
88. Ferretti G et al. Celiac disease, inflammation and oxidative damage: a nutrigenetic approach. Nutrients. Apr 2012;4(4):243-257.
89. Lee SA et al. DHA and EPA down-regulate COX-2 expression through suppression of NF-kappaB activity in LPS-treated human umbilical vein endothelial cells. Korean J Physiol Pharmacol. 2009;Aug;13:301-307.
90. Zapata-Gonzalez F et al. Human dendritic cell activities are modulated by the omega-3 fatty acid, docosahexaenoic acid, mainly through PPARgamma: RXR heterodimers: comparison with other polyunsaturated fatty acids. J Leukoc Biol. 2008;84:1172-1182.
91. Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. L-carnitine. Available at: http://lpi.oregonstate.edu/infocenter/othernuts/carnitine/. Last updated 4/2012a. Accessed 3/5/2015.
92. Ciacci C et al. L-carnitine in the treatment of fatigue in adult celiac disease patients: a pilot study. Dig Liver Dis. 2007;39:922-928.
93. Lerner A et al. Serum carnitine concentrations in coeliac disease. Gut. 1993;34:933-935.
94. Guandalini S et al. Celiac disease: a review. JAMA Pediatr 2014;168(3):272-278.
95. US NLM. Compound Summary for CID 124886. Glutathione. Pharmacology and Biochemistry. Available at: http://pubchem.ncbi.nlm.nih.gov/compound/glutathione#section=Pharmacology-and-Biochemistry. Accessed 3/5/2015.
96. Kidd PM. Glutathione: systemic protectant against oxidative and free radical damage. Alternative medicine review: a journal of clinical therapeutic. 1997;2(3):155-176.
97. Stojiljkovic V et al. Antioxidant status and lipid peroxidation in small intestinal mucosa of children with celiac disease. Clin Biochem. 2009;Sep;42(13-14):1432-7.
98. Stojiljkovic V et al. Glutathione redox cycle in small intestinal mucosa and peripheral blood of pediatric celiac disease patients. An Acad Bras Cienc. 2012;84(1):175-184.
99. Stojiljkovic V et al. Antioxidant enzymes, glutathione and lipid peroxidation in peripheral blood of children affected by coeliac disease. Ann Clin Biochem. 2007;44:537-543.
100. Richie JPJ et al. Randomized controlled trial of oral glutathione supplementation on body stores of glutathione. European journal of nutrition. 2014:1-13.
101. Lee S et al. Dietary L-cysteine improves the antioxidative potential and lipid metabolism in rats fed a normal diet. Bioscience, biotechnology, and biochemistry. 2013;77(7):1430-1434.
102. Sekhar RV et al. Deficient synthesis of glutathione underlies oxidative stress in aging and can be corrected by dietary cysteine and glycine supplementation. The American journal of clinical nutrition. Sep 2011;94(3):847-853.
103. Chen P et al. Dietary lipoic acid influences antioxidant capability and oxidative status of broilers. Int J Mol Sci. 2011;12(12):8476-8488.
104. Zavorsky GS et al. An open-label dose-response study of lymphocyte glutathione levels in healthy men and women receiving pressurized whey protein isolate supplements. Int J Food Sci Nutr. 2007;58(6):429-436.
105. Jiang X et al. [Effects of organic selenium supplement on glutathione peroxidase activities: a meta-analysis of randomized controlled trials]. Wei sheng yan jiu = Journal of hygiene research. Jan 2012;41(1):120-123.
106. Toouli J et al. Management of pancreatic exocrine insufficiency: Australasian Pancreatic Club recommendations. Med J Aust. 2010;193(8):461-7.
107. Malterre T. Digestive and nutritional considerations in celiac disease: could supplementation help? Alternative medicine review: a journal of clinical therapeutic. Sep 2009;14(3):247-257.
108. Evans KE et al. Pancreatic insufficiency in adult celiac disease: do patients require long-term enzyme supplementation? Digestive diseases and sciences. Oct 2010;55(10):2999-3004.
109. Weizman Z et al. Treatment failure in celiac disease due to coexistent exocrine pancreatic insufficiency. Pediatrics. Dec 1987;80(6):924-926.
110. Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Curcumin. Available at: http://lpi.oregonstate.edu/infocenter/phytochemicals/curcumin/. Last updated 1/2009. Accessed 3/5/2005.
111. Gupta SC et al. Therapeutic roles of curcumin: lessons learned from clinical trials. AAPS J. 2013;Jan;15(1):195-218.
112. Aggarwal BB et al. Potential therapeutic effects of curcumin, the anti-inflammatory agent, against neurodegenerative, cardiovascular, pulmonary, metabolic, autoimmune and neoplastic diseases. Int J Biochem Cell Biol. 2009;41:40-59.
113. Brumatti LV et al. Curcumin and inflammatory bowel disease: potential and limits of innovative treatments. Molecules 2014;19:211270-21153.
114. DiRaimondo TR et al. Interferon gamma activates transglutaminase 2 via a phosphatidylinositol-3-kinase-dependent pathway: implications for celiac sprue therapy. J Pharmacol Exp Ther. 2012;341(1):104-114.
115. Nilsen EM et al. Gluten induces an intestinal cytokine response strongly dominated by interferon gamma in patients with celiac disease. Gastroenterology. 1998;Sep;115(3):551-63.
116. Fahey AJ et al. Curcumin modulation of IFN-beta and IL-12 signalling and cytokine induction in human T cells. Journal of cellular and molecular medicine. Sep-Oct 2007;11(5):1129-1137.
117. Sanmukhani J et al. Efficacy and safety of curcumin in major depressive disorder: a randomized controlled trial. Phytother Res. 2014;Apr;28(4):579-85.
118. Antony B et al. Bioavailability of BCM-95 CG (Biocurcumax), a novel bioenhanced preparation of curcumin. Indian J Pharm Sci. 2008;Jul-Aug;70(4):445-9.
119. Peters SL et al. Randomized clinical trial: gluten may cause depression in subjects with non-coeliac gluten sensitivity – an exploratory clinical study. 2014;May;39(10):1104-12.
120. Smith DF et al. Meta-analysis on anxiety and depression in adult celiac disease. Acta Psychiatr Scand. 2012;Mar;125(3):189-93.
121. Bushara KO. Neurologic presentation of celiac disease. Gastroenterology. 2005;128:S92-S97.
122. Carta MG et al. Association between panic disorder, major depressive disorder and celiac disease: a possible role of thyroid autoimmunity. J Psychosom Res. 2002;Sep;53(3):789-93.
123. Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Lipoic Acid. Available at: http://lpi.oregonstate.edu/infocenter/othernuts/la/. Last updated 1/2012b. Accessed 3/5/2015.
124. Packer L et al. Lipoic acid: energy metabolism and redox regulation of transcription and cell signaling. J Clin Biochem Nutr. 2011;Jan;48:26-32.
125. Jones W et al. Uptake recycling, and antioxidant actions of alpha-lipoic acid in endothelial cells. Free Radic Biol Med. 2002;33(1):83-93.
126. Kolgazi M et al. Alpha-lipoic acid modulates gut inflammation induced by trinitrobenzene sulfonic acid in rats. J Gastroenterol Hepatol. 2007;22:1859-1865.
127. Suh JH et al. (R)-alpha-lipoic acid reverses the age-related loss in GSH redox status in post-mitotic tissues: evidence for increased cysteine requirement for GSH synthesis. Arch Biochem Biophys. 2004;423:126-135.
128. Ammon HP. Boswellic acids in chronic inflammatory diseases. Planta Med. 2006;72:1100-1116.
129. de Vasconcelos MI, Tirapegui J. [Nutritional importance of glutamine]. Arquivos de gastroenterologia. Jul-Sep 1998;35(3):207-215.
130. Miller AL. Therapeutic considerations of L-glutamine: a review of the literature. Alternative medicine review: a journal of clinical therapeutic. Aug 1999;4(4):239-248.
131. Li N et al. Glutamine regulates Caco-2 tight junction proteins. Am J Physiol Gastrointest Liver Physiol. 2004;287:G726-G733.
132. Beutheu S et al. Glutamine and arginine improve permeability and tight junction protein expression in methotrexate-treated Caco-2 cells. Clinical Nutrition. 2013;32:863-869.
133. Sevastiadou S et al. The impact of oral glutamine supplementation on the intestinal permeability and incidence of necrotizing enterocolitis/septicemia in premature neonates. J Matern Fetal Neonatal Med. 2011;24(10):1294-1300.
134. Choi K et al. The effect of oral glutamine on 5-fluorouracil/leucovorin-induced mucositis/stomatitis assessed by intestinal permeability test. Clin Nutr. 2007;26:57-62.
135. Benjamin J et al. Glutamine and whey protein improve intestinal permeability and morphology in patients with Crohn’s disease: a randomized controlled trial. Dig Dis Sci. 2012;57:1000-1012.
136. Kuhn KS et al. Glutamine as indispensable nutrient in oncology: experimental and clinical evidence. European journal of nutrition. Jun 2010;49(4):197-210.
137. Fasano A. Zonulin and its regulation of intestinal barrier function: the biological door to inflammation, autoimmunity, and cancer. Physiological Reviews. 2011;Jan1;91(1):151-175.
138. Arrieta MC et al. Alterations in intestinal permeability. Gut. 2006;55:1512:1520.
139. Alt Med Rev. Therapeutic applications of whey protein. Alternative Medicine Review. 2008;13(4):341-347.
140. Vilela RM et al. High hydrostatic pressure enhances whey protein digestibility to generate whey peptides that improve glutathione status in CFTR-deficient lung epithelial cells. Mol Nutr Food Res. 2006;50:1013-1029.
141. Kent KD et al. Effect of whey protein isolate on intracellular glutathione and oxidant-induced cell death in human prostate epithelial cells. Toxicol In Vitro. 2003;17:27-33.
142. Middleton N et al. Whole blood and mononuclear cell glutathione response to dietary whey protein supplementation in sedentary and trained male human subjects. Int J Food Sci Nutr. 2004;55(2):131-141.
美国梅奥诊所
www.mayoclinic.org
美国腹腔疾病基金会
http://www.celiac.org
美国腹腔疾病支持协会
http://www.csaceliacs.org
加拿大腹腔疾病协会
http://www.celiac.ca
加拿大卫生部
http://www.hc-sc.gc.ca
免责声明和安全信息
英文名称:Celiac Disease,Celiac Sprue,Gluten-Sensitive Enteropathy
定义
乳糜泻,也称为麦胶性肠病,是发生在消化道的一种自体免疫性疾病。在乳糜泻患者中,吃含麸质的食物会损害小肠吸收营养的绒毛。因此,可影响到对许多营养物质的吸收。未经治疗的乳糜泻通常会导致营养不良,并可引起腹泻、便秘和皮疹等。麦胶性肠病与小麦过敏、非腹腔麸胶过敏是不同的。病因
乳糜泻是由吃麸质食物引起的。乳糜泻的病因尚不清楚,很可能与遗传因素有关。带有这种特定基因的人在接触面筋类食物后会出现这种症状。乳糜泻的确切病因尚不清楚。分析认为,其发病原因存在下列因素:
- 乳糜泻是由食用含谷蛋白(麸质)的食物和其他环境因素造成基因相互作用而引起的。
- 婴儿喂养方法、胃肠道感染和肠道细菌异常可能导致乳糜泻发生。
- 妊娠、分娩,以及病毒感染或严重情绪压力之后,乳糜泻可能被诱发,或首次变得活跃。
- 当机体免疫系统对麸质反应过度时,可破坏小肠上的绒毛细胞—从食物中吸收维生素、矿物质和其他营养成分。如果绒毛受损,无论吃多少食物,都无法获得足够营养。
- 一些基因变异增加了发病风险,但必须涉及到其他因素。
- 无反应性的乳糜泻:
- 据调查,多达30%的乳糜泻患者可能没有或无法维持对无麸质饮食的良好反应。这通常是由于食用了受到麸质污染的食物。因此,接受专业营养师知道非常重要。
- 非反应性乳糜泻患者可能具有其他病症,例如小肠细菌过度生长,镜下结肠炎、胰腺功能差、肠易激综合征或对二糖(乳糖和果糖)不耐受等。或者,他们可能患有顽固性乳糜泻。
- 顽固性乳糜泻:在极少数患者中,即使遵循严格的无麸质饮食,乳糜泻的肠道损伤仍然存在并导致大量吸收不良,这就是难治性乳糜泻。
- 医生可能推荐类固醇激素治疗仪减少肠道炎症,或使用免疫抑制药物。注意进行随访,以监测对治疗的反应。
风险因素
女性比男性患病风险跟高。其他可增加乳糜泻发生的危险因素,包括如下:
- 家庭成员患有腹腔疾病
- 患有其他自体免疫性疾病史,如:
- 1型糖尿病
- 甲状腺疾病
- 系统性红斑狼疮
- 类风湿性关节炎
- 疱疹样皮炎,与腹腔疾病相关的皮炎
- 非母乳喂养
症状
乳糜泻症状可表现很大差异,可能始于童年或者成年期,儿童往往与成人有不同的症状。如果大段的肠道没有损坏,症状可能不明显。营养不良可能是乳糜泻的最初征兆,结果也是最严重的。- 儿童乳糜泻的体征和症状可能包括如下:
- 胃肠道症状,如腹痛、腹泻、恶心和呕吐,食欲不振,大便多且异味
- 情绪症状,如烦躁、易怒,严重者甚至癫痫发作
- 身体症状,如矮小、发育不良,嘴角裂疮和口内溃疡
- 成人乳糜泻的体征和症状可能包括如下
- 胃肠道症状,如腹胀、胀气,腹泻、腹痛、便秘,大便油性、恶臭、浅色等
- 神经与情绪症状,如反复性头痛、周围神经敏感下降、焦虑和惊恐发作,以及精神难于集中、抑郁等
- 其他身体症状,如疲劳、皮疹,体重下降和关节炎,月经失调和不明原因的不孕症或流产,以及贫血等。
- 贫血,通常由缺铁引起
- 骨密度丧失或骨软化
- 瘙痒,皮肤疱疹性皮炎(疱疹样皮炎)
- 牙釉质损伤
- 口腔溃疡
- 头痛和疲劳
- 神经系统损伤,包括脚部和手部麻木和刺痛,可能的平衡问题和认知功能障碍
- 关节疼痛
- 脾功能减退(脾虚症)
- 胃酸反流和胃灼热
- 疱疹洋皮炎:源于小肠麸质不耐受,与小肠内膜的变化相关。通常发生在肘部、膝盖、躯干、头皮和臀部。
并发症
乳糜泻可能会导致如下并发症:- 营养不良、贫血:对小肠的损害意味着它不能吸收足够的营养,导致贫血和体重减轻。
- 儿童生长发育受阻:营养吸收不良所致。
- 钙和骨密度的丧失:钙和维生素D的吸收不良可能导致儿童骨质软化,并导致成人骨质疏松症。
- 不孕和流产:营养吸收不良如维生素D、钙和贫血等,会导致生殖问题。
- 乳糖不耐症:尽管不含麸质,但吃了含乳糖的乳制品后,小肠受损可能会导致腹痛和腹泻。
- 神经问题:有些乳糜泻患者可能会出现神经问题,如癫痫发作或周围神经病变(导致手脚的神经疾病)。
- 癌症:乳糜泻患者如果不食用无麸质饮食,会发生多种癌症,包括肠淋巴瘤和小肠癌等。
疗法
综合选项包括如下:调整饮食与生活方式
- 避免所有含麦麸的食物,如小麦、黑麦和大麦等,这包括绝大多数面包、面食、谷类和加工食品。
- 麸质,也称为麦胶蛋白,是常用的食品加工原料,存在于许多意想不到的食物和饮料中,应仔细阅读产品标签,包括如:
- 风味咖啡、冰淇淋
- 金枪鱼蔬菜肉汤、奶油蔬菜、蔬菜沙拉和沙拉酱等
- 许多其他产品
营养与草本综合干预
以下是基于循证医学和循证营养学有关文献综合的结果。
有助于防控乳糜泻的营养和草本补充剂,主要包括如下:
1.维生素D:
维生素D缺乏是乳糜泻的常见问题,与一系列肌肉骨骼疾病有关,包括骨痛、肌肉疾病(肌病)、骨密度损失、骨质减少和骨质疏松症1-3。一项研究发现,患有乳糜泻的64%的男性和71%的女性维生素D水平较低4。然而,重要的是,这项研究将维生素D的“缺乏”定义为低于10ng/mL的25-羟基维生素D,这远低于被认为是最佳的水平。此外,有证据表明,乳糜泻患者的血清维生素D水平随着年龄的增长而降低,即使在阳光明媚的气候下也会出现低水平5。一份病例报告描述了一名患有肌病且维生素D水平低于4ng/dL的乳糜泻患者。患者开始无麸质饮食,并开始补充维生素D,剂量为30万IU每两周一次。患者的症状在一周内得到缓解3。
补充维生素D和钙,以及无麸质饮食,已被证明可以显著改善乳糜泻患者的骨骼状况6,7,2,并且广泛建议乳糜泻病人补充维生素D或钙8,9,2。维生素D除了在肌肉骨骼系统中的作用外,还有许多其他功能,特别是调节免疫系统和抗炎作用。来自实验室研究的数据为维生素D在减少人类全身炎症和预防自身免疫性疾病方面的潜力提供了证据10-12。具体而言,维生素D抑制促炎细胞因子(如干扰素γ和TNF-α)的分泌,同时促进抗炎细胞因子(包括IL-4、IL-5和IL-10)的产生13。
25-羟基维生素D的血液测试可以确定基线水平,并监测补充维生素D和无麸质饮食引起的水平变化。
2.钙:
在乳糜泻中,肠道内壁的损伤通常会导致钙吸收不良和血清钙水平下降2,14,15,8,然后导致骨质流失、骨质减少、骨质疏松和骨折风险增加16,17。在无麸质饮食中,钙水平和骨密度会改善,但只有在一到两年后才会提高18,19。然而,无麸质饮食可能钙含量低,并可能导致缺钙20-22和随后的低骨密度。因此,补充钙对乳糜泻患者很重要2,8,9,23。
3.铁:
缺铁性贫血是乳糜泻最常见的肠外症状之一18,24;也可能出现缺铁性贫血。尽管缺铁通常可以通过无麸质饮食来纠正,但在小肠内壁愈合后,铁水平的恢复可能需要6-12个月18。缺铁性血液测试,包括全血细胞计数和铁蛋白水平,在诊断中很重要25,补铁被认为是乳糜泻患者必要护理的一部分,部分原因是无麸质饮食可能导致营养不足20,26,27。
4.维生素B族:
缺乏叶酸和维生素B12和维生素B6在乳糜泻中很常见28-30,18,补充这些B族维生素以及烟酸(维生素B3)和维生素B2(核黄素),被认为是乳糜泻患者常规护理的重要组成部分31,32,27。在一项研究中,叶酸和5’-磷酸吡哆醛(维生素B6的活性形式)分别在37%和20%的成年乳糜泻患者中缺乏,这些患者已经无麸质饮食10年,并且活组织检查证明他们已经康复。在这项研究的参与者中,乳糜泻患者的平均每日叶酸和维生素B12摄入量显著低于对照组33。
同型半胱氨酸是一种氨基酸衍生物,可损伤血管内壁并促进动脉粥样硬化疾病34,35。同型半胱氨酸升高通常归因于缺乏维生素B12、B6和叶酸,而这些是同型半胱氨酸代谢所必需的36,37。几项研究表明,乳糜泻患者血液中同型半胱氨酸水平升高38,39。一项研究表明,与不使用维生素补充剂的患者相比,服用维生素补充剂的乳糜泻患者血液中叶酸、维生素B6和B12水平较高,同型半胱氨酸水平较低31。叶酸和维生素B12缺乏会导致维生素B缺乏性贫血,这在乳糜泻中很常见,高达34%的未经治疗的患者会出现这种情况40,41。
5.镁:
镁是人体内数百种酶促反应的必需矿物质42。镁也是维生素D和钙的适当代谢所必需的,因此也是骨骼健康所必需的43,44。
镁缺乏在乳糜泻中的重要性早在上世纪60年代首次被认识到,当时一份详细的病例报告显示,一名年轻成年乳糜泻患者在开始无麸质饮食后,镁的吸收和状态显著改善45。另一项研究调查了患有乳糜泻的儿童和青少年的镁状况。虽然所有患有典型乳糜泻(有吸收不良)的患者都缺乏镁,但只有五分之一的无麸质饮食患者(没有吸收不良)和五分之一患有无症状乳糜泻患者(没有吸收不良)的镁含量低46。后来的一项研究检测了41名患有乳糜泻或正常肠绒毛的儿童和青少年的镁,这些儿童和青少年平均11年无麸质饮食;28名未经治疗的非典型乳糜泻患者和8名对照组也被纳入研究。研究人员发现,通过红细胞镁水平评估,14.6%的无麸质饮食患者和25%的未经治疗的患者缺乏镁47。
一项对23名成年乳糜泻患者的早期研究发现,尽管他们没有症状(没有吸收不良),而且都在无麸质饮食中,但他们的细胞内镁水平都有所下降。这些人的镁治疗导致红细胞镁和骨密度增加,这表明镁缺乏可能在乳糜泻患者骨质疏松症的发展中发挥作用48。筛查镁的状况和缺乏,以及补充镁和增加饮食,被认为是乳糜泻患者护理的重要方面18,25,27。
6.锌:
锌是一种重要的营养矿物质,在体内300多种不同的酶促反应中起催化作用49。锌缺乏在未经治疗的乳糜泻中很常见,而且这种缺乏并不总是通过无麸质饮食来纠正;一些研究认为补锌是乳糜泻治疗的必要组成部分18,27,20。缺锌可能是乳糜泻生长、免疫功能、伤口愈合不良和皮肤问题的罪魁祸首。
一项对未经治疗和治疗的乳糜泻患者的研究综述发现,通过血浆或血清锌测量,锌缺乏的患病率显著。11项针对未经治疗的患有乳糜泻的成人和儿童的独立研究发现,高达100%的患者缺锌。9项针对乳糜泻患者的无麸质饮食研究中,有三项长达三个月至十年,发现20-40%的患者持续缺锌。仅在无麸质饮食中,缺锌通常需要一整年的时间才能解决,有人建议监测乳糜泻患者的血清锌水平18,32,50。
7.维生素E:
维生素E是一种重要的脂溶性维生素和自由基清除剂,在保护细胞膜免受氧化损伤方面发挥重要作用51。几项研究报告称,乳糜泻患者的维生素E水平较低50,52,53。维生素E缺乏也与乳糜泻神经症状的发展有关53-55。补充维生素E和无麸质饮食已被证明可以改善乳糜泻中维生素E缺乏引起的神经损伤53-55。
8.维生素A:
维生素A是一种脂溶性营养素,乳糜泻患者可能因吸收不良而缺乏维生素A。维生素A对正常免疫功能、视力和基因表达很重要56。乳糜泻维生素A缺乏症的治疗方法是无麸质饮食和补充27。
一项针对新诊断的乳糜泻患者的研究发现,其中7.5%的患者缺乏维生素A,而健康对照组则没有40。一项案例研究重申了维生素A的重要性,即使在控制性乳糜泻病例中也是如此。一名64岁的男性,经活检证实腹腔疾病在无麸质饮食中得到控制,最近出现腹泻,一只眼睛发红和模糊。在用药物治疗他的眼部症状的三周里,他的病情恶化了。维生素A的测试显示出严重缺乏。患者接受了含有100 ,000IU的维生素A,并且在一周内观察到视力和眼睛状况的显著改善。该患者的随访包括定期维生素A治疗57。
9.维生素K:
维生素K是脂溶性维生素之一,在乳糜泻中会因吸收不良而缺乏27,58,59。维生素K缺乏会导致容易出现瘀伤,这是乳糜泻的一种非典型症状;维生素K缺乏也已被证明会增加乳糜泻患者出现血栓和出血事件的风险60-63。在2012年一项针对患有乳糜泻的儿童和青少年的研究中,对维生素K的饮食摄入量进行了评估。在诊断时,41%的患者的摄入量低于推荐量的一半,而在无麸质饮食一年后,31%的患者的摄入仍然不足58。
已发现患有症状性和无症状性乳糜泻的儿童在诊断时骨量减少58,64,65。维生素K增强钙代谢,因此在骨骼健康中发挥重要作用66,67。由于钙、维生素K和D的饮食摄入通常不足,包括无麸质饮食的患者,因此建议在诊断为乳糜泻时常规补充维生素K58。
10.硒:
硒是谷胱甘肽过氧化物酶的组成部分,谷胱甘肽过氧化物酶在细胞抗氧化防御中发挥重要作用68。在一项针对30名患有乳糜泻的儿童的研究中,80%的儿童血清硒水平低于正常范围69。硒对甲状腺激素调节至关重要,乳糜泻中的硒缺乏与自身免疫性甲状腺疾病的风险增加有关70,71。
11.益生菌:
益生菌是定居在肠道中的有益微生物,在肠道健康中发挥着重要作用;在患有乳糜泻的患者中,这些生物体的平衡似乎紊乱了72,73。在一项为期三周的随机对照试验中,在未经治疗的乳糜泻患者中补充婴儿双歧杆菌NLS超级菌株可改善消化不良、便秘和胃酸倒流74。
目前正在开发各种益生菌菌株,专门针对乳糜泻的免疫和肠道损伤机制。乳双歧杆菌菌株已被证明可以保护培养的肠道上皮细胞免受面筋诱导的损伤,可能是通过阻断面筋诱导的肠道通透性增加75。此外,在一项实验室研究中,八种不同益生菌菌株的混合物(ACTIAL益生菌专有品牌VSL#3),包括几种乳酸菌和双歧杆菌,被证明可以广泛预消化小麦粉中的面筋蛋白。当乳糜泻患者的肠道活检样本暴露于这些预先消化的面筋蛋白时,免疫细胞的募集减少。研究人员得出结论,益生菌配方降低或消除了小麦面筋的毒性76。
12.维生素C:
维生素C是一种水溶性自由基清除剂,对全身组织的结构完整性也至关重要77。在一项实验中,用添加和不添加维生素C的麸质醇溶蛋白组分对乳糜泻患者的培养肠活检进行攻击。向培养细胞中添加维生素C降低了促炎细胞因子TNF-α、IL-6和干扰素γ的分泌。维生素C还完全抑制促炎细胞因子IL-15的分泌,IL-15与乳糜泻中发生的损伤密切相关78,79。
13.欧米伽3脂肪酸:
ω-3脂肪酸具有重要的生物抗炎作用80,81。当通过食物或补充剂摄入时,ω-3脂肪酸与更具促炎性的ω-6脂肪酸花生四烯酸竞争,以结合到富含脂质的细胞膜中82-84。
一项对新诊断为乳糜泻的成年人血清脂肪酸组成的研究发现,ω-3脂肪酸的浓度明显低于对照组。在无麸质饮食一年并获得临床缓解后,ω-3脂肪酸水平增加,但仍远低于对照值85。一项针对患有乳糜泻和1型糖尿病的儿童的研究发现,他们的ω-3脂肪酸水平(包括EPA和DHA)显著低于对照组86。
一项使用肠上皮细胞培养物的研究支持口服补充DHA在减少乳糜泻肠道炎症方面的潜在益处。作为对麸质引发的炎症的反应,肠细胞释放花生四烯酸。而DHA被证明可以阻断花生四烯酸的释放以及由此产生的炎症级联反应,后者促进疾病的发展87,88。ω-3脂肪酸减少炎症的另一种机制是通过抑制核因子κB(NF-ĸB),一种促炎介质89。DHA还刺激一种名为PPARγ的细胞受体,该受体激活减少促炎细胞因子产生的基因90。
14.L肉碱:
L-肉碱是一种在人体内由赖氨酸和蛋氨酸制成的化合物,在饮食中也主要从肉类、鱼类和乳制品等动物性食品中获得91。在一项随机对照试验中,连续六个月每天补充2g L-肉碱可显著改善患有乳糜泻的成年人的疲劳。L-肉碱的剂量是安全且耐受性良好的92)。
L-肉碱需要将长链脂肪酸穿梭到线粒体(细胞“能量站”)中,以产生肌肉能量。乳糜泻的肠道损伤导致吸收表面积减少,增加了L-肉碱缺乏的风险。在一项研究中,未经治疗的乳糜泻患者的血清L-肉碱浓度较低,在无麸质饮食后恢复正常93。另一项研究发现患有乳糜泻的儿童血清肉碱水平较低69。L-肉碱缺乏可能会导致疲劳、虚弱和体重增加困难,这些都是乳糜泻的常见症状93,94。
15.谷胱甘肽:
谷胱甘肽是人体对抗破坏性自由基最强大的防御系统之一,并对肝脏的解毒功能至关重要95,96。几项研究报告称,与对照组相比,乳糜泻患者肠道内壁97,98和外周血98,99中的谷胱甘肽浓度显著降低。结果,肠内膜中脂质过氧化物或酸腐脂肪的浓度增加。脂质过氧化物可促进组织中的一系列氧化损伤,导致肠道中的组织损伤,这是乳糜泻的特征,并增加乳糜泻患者患癌症的风险97-99。
通过直接补充谷胱甘肽100或使用生理前体如半胱氨酸101,102和N-乙酰半胱氨酸95,96,以及α-硫辛酸、硒和乳清蛋白103-105,可以增加人体的谷胱甘肽储备。
16.消化酶(胰酶):
胰腺外分泌功能不全是一种没有足够的消化酶分泌到小肠中的情况106,在对无麸质饮食没有反应的乳糜泻患者中相对常见25。小肠受损可能会抑制胰腺分泌消化酶的能力。一项研究发现,即使在无麸质饮食后腹泻仍持续的病例中,胰腺功能不全的实验室症状发生率明显更高。接受胰酶治疗的患者症状减轻了75%。另一项研究发现,小肠绒毛萎缩的严重程度与胰腺外分泌功能不全的实验室体征显著相关。在一项研究中,超过一半的乳糜泻患者出现胰腺酶功能不全,而在无麸质饮食一年后恢复正常107-109。
17.其他支持:
以下干预措施尚未在乳糜泻的背景下进行彻底研究,但可能有助于支持肠道健康或对抗炎症反应或细胞代谢紊乱,这被认为是乳糜泻几种表现的病理基础。
- 姜黄素:姜黄素是姜黄中的主要多酚化合物,姜黄是印度阿育吠陀医学中使用了几个世纪的植物110。姜黄素具有强大的抗炎和抗氧化特性,并在实验室、动物和人类模型中证明了其抵御多种疾病的能力,包括自体免疫疾病和IBD111-113。
与克罗恩病和溃疡性结肠炎一样,乳糜泻是一种炎症性自体免疫性疾病25。在乳糜泻中,炎症介质干扰素γ促进小肠通透性增强和损伤114,115。姜黄素已被证明可以抑制干扰素γ,这表明姜黄素可能对乳糜泻有益116。
有趣的是,姜黄素还可能改善乳糜泻的心理表现和非乳糜泻面筋敏感性。在一项随机对照试验中,发现一种生物利用率高的姜黄素与标准抗抑郁药物氟西汀(百忧解)在治疗重度抑郁症方面同样有效117,118。由于抑郁症和乳糜泻的其他心理表现以及非乳糜泻面筋敏感性正越来越频繁地被认识到119-122,这是姜黄素的另一个益处,可能对这些面筋相关疾病有价值。
- α-硫辛酸:α-硫辛酸是一种天然存在的化合物,主要参与细胞线粒体内的能量生产。它也是一种强大的自由基清除剂123。此外,α-硫辛酸还能再生其他自由基清除剂,如谷胱甘肽、维生素C、维生素E和辅酶Q10103,124,125。
α-硫辛酸增加谷胱甘肽水平的能力在乳糜泻中很重要,乳糜泻的谷胱甘肽水平较低很常见97,99。在动物模型中,饮食中补充α-硫辛酸显著提高了细胞谷胱甘肽浓度和谷胱甘肽过氧化物酶的活性,谷胱甘肽过氧化物酶是一种关键的谷胱甘肽依赖性抗氧化酶。与对照组相比,氧化脂质(过氧化)水平也显著降低103。其他研究支持α-硫辛酸加速谷胱甘肽合成的潜力124,126,127。在一种肠道炎症(乳糜泻疾病的一个特征)的啮齿动物模型中,用α-硫辛酸治疗大鼠显著减少了对肠道衬里的几种损伤126。
- 乳香:近年来,在欧洲部分地区,从乳香树脂和其他乳香属物种中提取的制剂,已成为治疗包括慢性肠道疾病在内的各种慢性炎症的热门药物。临床研究表明,乳香治疗自身免疫性肠病(克罗恩病和溃疡性结肠炎)是安全有效的。乳香酸是乳香的主要活性成分,通过抑制涉及5-脂氧合酶(5-LOX)、白三烯和TNF-α的促炎途径发挥抗炎作用128。类似地,乳香可能有助于减少乳糜泻疾病的肠道炎症。
- 谷氨酰胺:谷氨酰胺是一种氨基酸,对胃肠道内壁特别重要,尤其是在压力和疾病期间。谷氨酰胺是体内最丰富的氨基酸,也是肠道细胞的关键燃料。事实上,口服谷氨酰胺能够增加肠道绒毛的高度,帮助肠道内壁的细胞增殖,并保持肠道内壁的完整性,防止过度渗透129,130。作为将肠细胞“粘合”在一起的紧密连接蛋白的调节因子131,谷氨酰胺已被证明在各种临床条件下对肠道屏障功能(通透性)发挥积极作用132-134。在2012年一项针对处于缓解期的克罗恩病患者的随机对照试验中,口服谷氨酰胺以0.23克/磅体重持续两个月,显著改善了肠道通透性和粘膜完整性135。
补充谷氨酰胺治疗乳糜泻尚待研究。然而,由于补充谷氨酰胺已被证明可以安全地增强和维持肠道屏障功能,其在乳糜泻引起的通透性增加中的应用是一个具有重大科学意义的领域134-138。
- 乳清蛋白:谷胱甘肽是保护肠粘膜(肠道内壁)免受氧化损伤的防御机制的组成部分97。乳清蛋白是合成谷胱甘肽所需的半胱氨酸的丰富来源139。几项研究表明,乳清蛋白对提高各种类型细胞中的谷胱甘肽水平非常有效104,140-142。
此外,在2012年一项针对克罗恩病患者的研究中,乳清蛋白显著降低了肠道通透性135。乳清对肠道完整性的这种益处可能对乳糜泻有用,乳清蛋白可以很容易地作为高质量蛋白质和谷胱甘肽前体的来源纳入无麸质饮食中。
更多可点击其个性化综合干预方案如下:
- 乳糜泻管理(防营养不良)
- 乳糜泻管理(防止骨松症)
- 乳糜泻管理(维持消化功能)
- 乳糜泻管理(抗自体免疫)
- 乳糜泻管理(改善病理基础)
- 乳糜泻防控(儿童)
- 乳糜泻防控(青少年)
- 乳糜泻防控(青年)
- 乳糜泻防控(中年)
- 乳糜泻防控(老年)
以及参阅本网如下专文的相关内容:
医疗干预
治疗乳糜泻需要面临如下问题:
- 首先实施终身的、无麸质饮食是腹腔疾病的唯一治疗法,并且非常快速、有效。
- 因乳糜泻损害的小肠绒毛可能需要数月或数年才能愈合。
- 如果肠道损伤不愈合,可能需要通过静脉给予营养补充。
- 被延误的生长发育和牙齿变色可能是永久性存在的。
- 麸质(谷蛋白、麦胶蛋白)是运用广泛的食品原料,存在于许多食物中,现实中很难避免。此外,在许多食品添加剂,甚至营养保健品、药品等中,作为辅料的谷蛋白都可能存在。
- 及时筛查和补充营养。
- 医生可能建议服用补充剂,包括维生素D、钙、叶酸、维生素B12、铁和锌等。如果消化道吸收不良,可能需要注射补给。服用补充剂时,要注意不含麸质。
- 医学随访、定期体检,以确保获得足够的营养,包括骨密度测试。
- 控制肠道炎症药物:当小肠严重受损,可能要使用类固醇控制炎症。
- 疱疹样皮炎:使用外用药物,结合严格的无麸质饮食。
- 顽固性腹腔疾病:由于对无麸质饮食无效、难于控制,症状可能变得严重,或者病情反复。因此,可能需要接受医学团队评估和治疗。不过,目前尚无经证实的治疗方法。
预防
因为病因还不清楚,目前还没有预防乳糜泻的指南。如果孩子患腹腔疾病的风险增加,日常生活中要尽量食用无麸质食品。
越早吃无麸质饮食,对肠道的损伤就越小。
参考文献:
1. Mulder CJ et al. Celiac disease presenting as severe osteopenia. Hawaii medical journal. Nov 2011;70(11):242-244.
2. Rastogi A et al. Celiac disease: a missed cause of metabolic bone disease. Indian J Endocrinol Metab. 2012;Sep-Oct;16(5):780-785.
3. Karaahmet OZ et al. Myopathy related to vitamin D deficiency in patient with celiac disease. Muscle & nerve. Jul 2014;50(1):147-148.
4. Kemppainen T et al. Osteoporosis in adult patients with celiac disease. Bone. 1999;24(3):249-255.
5. Lerner A et al. The clinical significance of 25OH-vitamin D status in celiac disease. Clinic Rev Allerg Immunol. 2012;42:322-330.
6. Duerksen DR. Dramatic effect of vitamin D supplementation and a gluten-free diet on bone mineral density in a patient with celiac disease. J Clin Densitom. 2012;Jan-Mar;15(1):120-123.
7. Bianchi ML, Bardella MT. Bone in celiac disease. Osteoporos Int. 2008;19:1705-1716.
8. Szymczak J et al. Low bone mineral density in adult coeliac disease. Endokrynol Pol. 2012;63(4):270-6.
9. Lucendo AJ et al. Bone mineral density in adult coeliac disease: an updated review. Rev Esp Enferm Dig. 2013;Mar;105(3):154-62.
10. Richards JB et al. Higher serum vitamin D concentrations are associated with longer leukocyte telomere length in women. The American journal of clinical nutrition. Nov 2007;86(5):1420-1425.
11. Kriegel MA et al. Does vitamin D affect risk of developing autoimmune disease?: a systematic review. Semin Arthritis Rheum. 2011;40:512-531.
12. Agmon-Levin N et al. Vitamin D in systemic and organ-specific autoimmune diseases. Clinic Rev Allerg Immunol. 2013;45:256-266.
13. Prietl B et al. Vitamin D and immune function. Nutrients. 2013;5:2502-2521.
14. Pantaleoni S et al. Bone mineral density at diagnosis of celiac disease and after 1 year of gluten-free diet. Scientific World Journal. 2014;2014:173082.
15. Babar MI et al. Celiac disease and celiac crisis in children. J Coll Physicians Surg Pak. 2011;Aug;21(8):487-90.
16. AGA. American Gastroenterological medical position statement: celiac sprue. Gastroenterology. 2001;120(6):1522-5.
17. Pantaleoni S et al. Bone mineral density at diagnosis of celiac disease and after 1 year of gluten-free diet. Scientific World Journal. 2014;2014:173082.
18. Caruso R et al. Appropriate nutrient supplementation in celiac disease. Ann Med. 2013;Dec;45(8):522-31.
19. Molteni N et al. Intestinal calcium absorption as shown by stable strontium test in celiac disease before and after gluten-free diet. Am J Gastroenterol. 1995; Nov;90(11):2025-8.
20. Kupper C. Dietary guidelines and implementation for celiac disease. Gastroenterology. Apr 2005;128(4 Suppl 1):S121-127.
21. UCMC. The University of Chicago Celiac Disease Center. Jump start your gluten-free diet. Available at: http://www.cureceliacdisease.org/living-with-celiac/resources/jump-start-your-gluten-free-diet-ebook. Updated 2013. Accessed 10/17/2014.
22. Blazina S et al. Bone mineral density and importance of strict gluten-free diet in children and adolescents with celiac disease. Bone. 2010;Sep;47(3):598-603.
23. Mancini LA et al. Celiac disease and the athlete. Curr Sports Med Rep. 2011;Mar-Apr;10(2):105-8.
24. Horowitz S. Celiac Disease: New Directions in Diagnosis, Treatment, and Prevention. Alternative and Complementary Therapies. 2011;17(2):92-98.
25. Ferri FF. Ferri's Clinical Advisor. Celiac Disease. Available at: www.clinicalkey.com. Copyright 2015. Accessed 2/3/2015.
26. Thompson T. Folate, iron, and dietary fiber contents of the gluten-free diet. Journal of the American Dietetic Association. Nov 2000;100(11):1389-1396.
27. Lidums I et al. First Consult. Celiac disease. Available at: www.clinicalkey.com. Copyright 2015 by Elsevier Inc.
28. Dickey W. Low serum vitamin B12 is common in coeliac disease and is not due to autoimmune gastritis. Eur J Gastroenterol Hepatol. 2002;14(4):425-427.
29. Haapalahti M et al. Nutritional status in adolescents and young adults with screen-detected celiac disease. J Pediatr Gastroenterol of Nutr. 2005;May;40:566-570.
30. Tikkakoski S et al. Undiagnosed coeliac disease and nutritional deficiencies in adults screened in primary health care. Scand J Gastroenterol. 2007;Jan;’42(1):60-65.
31. Hadithi M et al. Effect of B vitamin supplementation on plasma homocysteine levels in celiac disease. World J Gastroenterol. 2009;Feb 28;15(8):955-960.
32. Wierdsma NJ, van Bokhorst-de van der Schueren MA, Berkenpas M, et al. Vitamin and mineral deficiencies are highly prevalent in newly diagnosed celiac disease patients. Nutrients. 2013;Sep 30;5(10):3975-92.
33. Hallert C et al. Evidence of poor vitamin status in coeliac patients on a gluten-free diet for 10 years. Aliment Pharmacol Ther. 2002;16:1333-1339.
34. Eren E et al. Homocysteine, Paraoxonase-1 and Vascular Endothelial Dysfunction: Omnibus viis Romam Pervenitur. Journal of clinical and diagnostic research: JCDR. Sep 2014;8(9):Ce01-04.
35. Pushpakumar S et al. Endothelial dysfunction: the link between homocysteine and hydrogen sulfide. Current medicinal chemistry. 2014;21(32):3662-3672.
36. Higdon J. Linus Pauling Institute. Micronutrient Information Center. Vitamin B12. Available at: http://lpi.oregonstate.edu/infocenter/vitamins/vitaminB12/. Last updated 1/2014a. Accessed 3/5/2015.
37. Higdon J. Linus Pauling Institute. Micronutrient Information Center. Vitamin B6. Available at: http://lpi.oregonstate.edu/infocenter/vitamins/vitaminB6/. Last updated 5/2014b. Accessed 3/5/2015.
38. Hallert C et al. Evidence of poor vitamin status in coeliac patients on a gluten-free diet for 10 years. Aliment Pharmacol Ther. 2002;16:1333-1339.
39. Dickey W et al. Homocysteine and related B-vitamin status in coeliac disease: effects of gluten exclusion and histological recovery. Scand J Gastroenterol. 2008;43:682-688.
40. Wierdsma NJ et al. Vitamin and mineral deficiencies are highly prevalent in newly diagnosed celiac disease patients. Nutrients. 2013;Sep 30;5(10):3975-92.
41. Halfdanarson TR et al. Hematologic manifestations of celiac disease. Blood. 2007 Jan 15;109(2):412-421.
42. Higdon J. Linus Pauling Institute. Micronutrient Information Center. Micronutrient Information Center. Magnesium. Available at: http://lpi.oregonstate.edu/infocenter/minerals/magnesium/. Last updated 10/2013a. Accessed 2/11/2015.
43. Zofkova I et al. The relationship between magnesium and calciotropic hormones. Magnes Res. 1995;Mar;8(1):77-84.
44. Hardwick LL et al. Magnesium absorption: mechanisms and the influence of vitamin D, calcium and phosphate. The Journal of nutrition. Jan 1991;121(1):13-23.
45. Goldman AS et al. Magnesium deficiency in celiac disease. Pediatrics. Jun 1962;29:948-952.
46. Rujner J et al. Magnesium status in children and adolescents with celiac disease. Wiad Lek. 2001;54(5-6):277-285.
47. Rujner J et al. Magnesium status in children and adolescents with coeliac disease without malabsorption symptoms. Clinical nutrition (Edinburgh, Scotland). Oct 2004;23(5):1074-1079.
48. Rude RK et al. Magnesium deficiency: possible role in osteoporosis associated with gluten-sensitive enteropathy. Osteoporos Int. 1996;6(6):453-61.
49. Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Zinc. Available at: http://lpi.oregonstate.edu/infocenter/minerals/zinc/. Last updated 6/2013b. Accessed 3/5/2015.
50. Guevara Pacheco G et al. [Micronutrient deficiencies and celiac disease in Pediatrics]. Arch Argent Pediatr. 2014;Oct;112(5):457-63.
51. Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Vitamin E. Available at: http://lpi.oregonstate.edu/infocenter/vitamins/vitaminE/. Last updated 6/2008. Accessed 3/5/2015.
52. Odetti P et al. Oxidative stress in subjects affected by celiac disease. Free Radic Res. 1998;Jul;29(1):17-24.
53. Henri-Bhargava A et al. Neurologic impairment due to vitamin E and copper deficiencies in celiac disease. Neurology. 2008;Sep 9;71(11):860-1.
54. Kleopa KA et al. Reversible inflammatory and vacuolar myopathy with vitamin E deficiency in celiac disease. Muscle Nerve. 2005;31:260-265.
55. Mauro A et al. Cerebellar syndrome in adult celiac disease with vitamin E deficiency. Acta Neurol Scand. 1991;84:167-170.
56. Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Vitamin A. Available at: http://lpi.oregonstate.edu/infocenter/vitamins/vitaminA/. Last updated 11/2007a. Accessed 3/5/2015.
57. Alwitry A. Vitamin A deficiency in coeliac disease. The British Journal of Ophthalmology. 2000;84(9):1075-1075.
58. Mager DR et al. Vitamin D and K status influences bone mineral density and bone accrual in children and adolescents with celiac disease. Eur J Clin Nutr. 2012;66:488-495.
59. Kelly CP et al. ePocrates online. Celiac disease. Available at: https://online.epocrates.com/noFrame/showPage?method=diseases&MonographId=636. Last updated 4/11/2014. Accessed 2/3/2015.
60. Berthoux E et al. [Adult celiac disease with thrombosis: a case series of seven patients. Role of thrombophilic factors]. La Revue de medecine interne / fondee ... par la Societe nationale francaise de medecine interne. Oct 2011;32(10):600-604.
61. Chen CS et al. Coagulopathy due to celiac disease presenting as intramuscular hemorrhage. Journal of general internal medicine. Nov 2007;22(11):1608-1612.
62. Vaynshtein G et al. Celiac sprue presenting as severe hemorrhagic diathesis due to vitamin K deficiency. The Israel Medical Association journal : IMAJ. Dec 2004;6(12):781-783.
63. Lerner A et al. Hypercoagulability in celiac disease--an update. Autoimmunity reviews. Nov 2014;13(11):1138-1141.
64. Turner J et al. Prevalence of metabolic bone disease in children with celiac disease is independent of symptoms at diagnosis. J Pediatr Gastroenterol Nutr. 2009;49:589-593.
65. Rajani S et al. The changing frequency of celiac disease diagnosed at the Stollery Children’s Hospital. Can J Gastroenterol. 2010;24(2):109-112.
66. Zittermann A. Effects of vitamin K on calcium and bone metabolism. Current opinion in clinical nutrition and metabolic care. Nov 2001;4(6):483-487.
67. Booth SL. Roles for vitamin K beyond coagulation. Annu Rev Nutr. 2009;29:89-110.
68. Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Selenium. Available at: http://lpi.oregonstate.edu/infocenter/minerals/selenium/. Last updated 11/2007b. Accessed 3/5/2015.
69. Yuce A et al. Serum carnitine and selenium levels in children with celiac disease. Indian J Gastroenterol. 2004;May-June;23:87-88.
70. Stazi AV et al. Selenium deficiency in celiac disease: risk of autoimmune thyroid diseases. Minerva Med. 2008;Dec;99(6):643-53.
71. Stazi AV et al. Selenium status and over-expression of interleukin-15 in celiac disease and autoimmune thyroid diseases. Ann Ist Super Sanita. 2010;46(4):389-399.
72. Pozo-Rubio T et al. Immune development and intestinal microbiota in celiac disease. Clin Dev Immunol. 2012;2012:654143.
73. Round JL et al. The gut microbiota shapes intestinal immune responses during health and disease. Nat Rev Immunol. 2009;May;9:313-323.
74. Smecuol E et al. Exploratory, randomized, double-blind, placebo-controlled study on the effects of Bifidobacterium infantis natren life start strain super strain in active celiac disease. J Clin Gastroenterol. 2013;47:139-147.
75. Lindfors K et al. Live probiotic Bifidobacterium lactis bacteria inhibit the toxic effects induced by wheat gliadin in epithelial cell culture. Clin Exp Immunol. 2008;152:552-558.
76. De Angelis M et al. VSL#3 probiotic preparation has the capacity to hydrolyze gliadin polypeptides responsible for Celiac Sprue. Biochim Biophys Acta. 2006;Jan;1762(1):80-93.
77. Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Vitamin C. Available at: http://lpi.oregonstate.edu/infocenter/vitamins/vitaminC/#function. Last updated 11/2013c. Accessed 3/5/2015.
78. Bernardo D et al. Ascorbate-dependent decrease of the mucosal immune inflammatory response to gliadin in celiac disease patients. Allergol Immunopathol (Madr). 2012;40(1):3-8.
79. Gujral N et al. Celiac disease: Prevalence, diagnosis, pathogenesis and treatment. World J Gastroenterol. 2012;18(42):6036-6059.
80. Skulas-Ray AC. Omega-3 fatty acids and inflammation: A perspective on the challenges of evaluating efficacy in clinical research. Prostaglandins & other lipid mediators. 2015 Jan-Mar:116-117:104-11.
81. Calder PC. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochimica et biophysica acta. Apr 2015;1851(4):469-484.
82. Westphal C et al. CYP-eicosanoids – a new link between omega-3 fatty acids and cardiac disease? Prostaglandins Other Lipid Mediat. 2011;96:99-108.
83. Calder PC. Immunomodulation by omega-3 fatty acids. Prostaglandins Leukot Essent Fatty Acids. 2007;77:327-335.
84. Surette ME. The science behind dietary omega-3 fatty acids. Canadian Medical Association journal. Jan 15 2008;178(2):177-180.
85. Solakivi T et al. Serum fatty acid profile in celiac disease patients before and after a gluten-free diet. Scand J Gastrolenterol. 2009;44:826-830.
86. Tarnok A et al. Low n-3 Long-Chain Polyunsaturated Fatty Acids in Newly Diagnosed Celiac Disease in Children With Preexisting Type 1 Diabetes Mellitus. Journal of pediatric gastroenterology and nutrition. Feb 2015;60(2):255-258.
87. Vincentini O et al. Docosahexaenoic acid modulates in vitro the inflammation of celiac disease in intestinal epithelial cells via the inhibition of cPLA2. Clin Nutr. 2011;30:541-546.
88. Ferretti G et al. Celiac disease, inflammation and oxidative damage: a nutrigenetic approach. Nutrients. Apr 2012;4(4):243-257.
89. Lee SA et al. DHA and EPA down-regulate COX-2 expression through suppression of NF-kappaB activity in LPS-treated human umbilical vein endothelial cells. Korean J Physiol Pharmacol. 2009;Aug;13:301-307.
90. Zapata-Gonzalez F et al. Human dendritic cell activities are modulated by the omega-3 fatty acid, docosahexaenoic acid, mainly through PPARgamma: RXR heterodimers: comparison with other polyunsaturated fatty acids. J Leukoc Biol. 2008;84:1172-1182.
91. Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. L-carnitine. Available at: http://lpi.oregonstate.edu/infocenter/othernuts/carnitine/. Last updated 4/2012a. Accessed 3/5/2015.
92. Ciacci C et al. L-carnitine in the treatment of fatigue in adult celiac disease patients: a pilot study. Dig Liver Dis. 2007;39:922-928.
93. Lerner A et al. Serum carnitine concentrations in coeliac disease. Gut. 1993;34:933-935.
94. Guandalini S et al. Celiac disease: a review. JAMA Pediatr 2014;168(3):272-278.
95. US NLM. Compound Summary for CID 124886. Glutathione. Pharmacology and Biochemistry. Available at: http://pubchem.ncbi.nlm.nih.gov/compound/glutathione#section=Pharmacology-and-Biochemistry. Accessed 3/5/2015.
96. Kidd PM. Glutathione: systemic protectant against oxidative and free radical damage. Alternative medicine review: a journal of clinical therapeutic. 1997;2(3):155-176.
97. Stojiljkovic V et al. Antioxidant status and lipid peroxidation in small intestinal mucosa of children with celiac disease. Clin Biochem. 2009;Sep;42(13-14):1432-7.
98. Stojiljkovic V et al. Glutathione redox cycle in small intestinal mucosa and peripheral blood of pediatric celiac disease patients. An Acad Bras Cienc. 2012;84(1):175-184.
99. Stojiljkovic V et al. Antioxidant enzymes, glutathione and lipid peroxidation in peripheral blood of children affected by coeliac disease. Ann Clin Biochem. 2007;44:537-543.
100. Richie JPJ et al. Randomized controlled trial of oral glutathione supplementation on body stores of glutathione. European journal of nutrition. 2014:1-13.
101. Lee S et al. Dietary L-cysteine improves the antioxidative potential and lipid metabolism in rats fed a normal diet. Bioscience, biotechnology, and biochemistry. 2013;77(7):1430-1434.
102. Sekhar RV et al. Deficient synthesis of glutathione underlies oxidative stress in aging and can be corrected by dietary cysteine and glycine supplementation. The American journal of clinical nutrition. Sep 2011;94(3):847-853.
103. Chen P et al. Dietary lipoic acid influences antioxidant capability and oxidative status of broilers. Int J Mol Sci. 2011;12(12):8476-8488.
104. Zavorsky GS et al. An open-label dose-response study of lymphocyte glutathione levels in healthy men and women receiving pressurized whey protein isolate supplements. Int J Food Sci Nutr. 2007;58(6):429-436.
105. Jiang X et al. [Effects of organic selenium supplement on glutathione peroxidase activities: a meta-analysis of randomized controlled trials]. Wei sheng yan jiu = Journal of hygiene research. Jan 2012;41(1):120-123.
106. Toouli J et al. Management of pancreatic exocrine insufficiency: Australasian Pancreatic Club recommendations. Med J Aust. 2010;193(8):461-7.
107. Malterre T. Digestive and nutritional considerations in celiac disease: could supplementation help? Alternative medicine review: a journal of clinical therapeutic. Sep 2009;14(3):247-257.
108. Evans KE et al. Pancreatic insufficiency in adult celiac disease: do patients require long-term enzyme supplementation? Digestive diseases and sciences. Oct 2010;55(10):2999-3004.
109. Weizman Z et al. Treatment failure in celiac disease due to coexistent exocrine pancreatic insufficiency. Pediatrics. Dec 1987;80(6):924-926.
110. Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Curcumin. Available at: http://lpi.oregonstate.edu/infocenter/phytochemicals/curcumin/. Last updated 1/2009. Accessed 3/5/2005.
111. Gupta SC et al. Therapeutic roles of curcumin: lessons learned from clinical trials. AAPS J. 2013;Jan;15(1):195-218.
112. Aggarwal BB et al. Potential therapeutic effects of curcumin, the anti-inflammatory agent, against neurodegenerative, cardiovascular, pulmonary, metabolic, autoimmune and neoplastic diseases. Int J Biochem Cell Biol. 2009;41:40-59.
113. Brumatti LV et al. Curcumin and inflammatory bowel disease: potential and limits of innovative treatments. Molecules 2014;19:211270-21153.
114. DiRaimondo TR et al. Interferon gamma activates transglutaminase 2 via a phosphatidylinositol-3-kinase-dependent pathway: implications for celiac sprue therapy. J Pharmacol Exp Ther. 2012;341(1):104-114.
115. Nilsen EM et al. Gluten induces an intestinal cytokine response strongly dominated by interferon gamma in patients with celiac disease. Gastroenterology. 1998;Sep;115(3):551-63.
116. Fahey AJ et al. Curcumin modulation of IFN-beta and IL-12 signalling and cytokine induction in human T cells. Journal of cellular and molecular medicine. Sep-Oct 2007;11(5):1129-1137.
117. Sanmukhani J et al. Efficacy and safety of curcumin in major depressive disorder: a randomized controlled trial. Phytother Res. 2014;Apr;28(4):579-85.
118. Antony B et al. Bioavailability of BCM-95 CG (Biocurcumax), a novel bioenhanced preparation of curcumin. Indian J Pharm Sci. 2008;Jul-Aug;70(4):445-9.
119. Peters SL et al. Randomized clinical trial: gluten may cause depression in subjects with non-coeliac gluten sensitivity – an exploratory clinical study. 2014;May;39(10):1104-12.
120. Smith DF et al. Meta-analysis on anxiety and depression in adult celiac disease. Acta Psychiatr Scand. 2012;Mar;125(3):189-93.
121. Bushara KO. Neurologic presentation of celiac disease. Gastroenterology. 2005;128:S92-S97.
122. Carta MG et al. Association between panic disorder, major depressive disorder and celiac disease: a possible role of thyroid autoimmunity. J Psychosom Res. 2002;Sep;53(3):789-93.
123. Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Lipoic Acid. Available at: http://lpi.oregonstate.edu/infocenter/othernuts/la/. Last updated 1/2012b. Accessed 3/5/2015.
124. Packer L et al. Lipoic acid: energy metabolism and redox regulation of transcription and cell signaling. J Clin Biochem Nutr. 2011;Jan;48:26-32.
125. Jones W et al. Uptake recycling, and antioxidant actions of alpha-lipoic acid in endothelial cells. Free Radic Biol Med. 2002;33(1):83-93.
126. Kolgazi M et al. Alpha-lipoic acid modulates gut inflammation induced by trinitrobenzene sulfonic acid in rats. J Gastroenterol Hepatol. 2007;22:1859-1865.
127. Suh JH et al. (R)-alpha-lipoic acid reverses the age-related loss in GSH redox status in post-mitotic tissues: evidence for increased cysteine requirement for GSH synthesis. Arch Biochem Biophys. 2004;423:126-135.
128. Ammon HP. Boswellic acids in chronic inflammatory diseases. Planta Med. 2006;72:1100-1116.
129. de Vasconcelos MI, Tirapegui J. [Nutritional importance of glutamine]. Arquivos de gastroenterologia. Jul-Sep 1998;35(3):207-215.
130. Miller AL. Therapeutic considerations of L-glutamine: a review of the literature. Alternative medicine review: a journal of clinical therapeutic. Aug 1999;4(4):239-248.
131. Li N et al. Glutamine regulates Caco-2 tight junction proteins. Am J Physiol Gastrointest Liver Physiol. 2004;287:G726-G733.
132. Beutheu S et al. Glutamine and arginine improve permeability and tight junction protein expression in methotrexate-treated Caco-2 cells. Clinical Nutrition. 2013;32:863-869.
133. Sevastiadou S et al. The impact of oral glutamine supplementation on the intestinal permeability and incidence of necrotizing enterocolitis/septicemia in premature neonates. J Matern Fetal Neonatal Med. 2011;24(10):1294-1300.
134. Choi K et al. The effect of oral glutamine on 5-fluorouracil/leucovorin-induced mucositis/stomatitis assessed by intestinal permeability test. Clin Nutr. 2007;26:57-62.
135. Benjamin J et al. Glutamine and whey protein improve intestinal permeability and morphology in patients with Crohn’s disease: a randomized controlled trial. Dig Dis Sci. 2012;57:1000-1012.
136. Kuhn KS et al. Glutamine as indispensable nutrient in oncology: experimental and clinical evidence. European journal of nutrition. Jun 2010;49(4):197-210.
137. Fasano A. Zonulin and its regulation of intestinal barrier function: the biological door to inflammation, autoimmunity, and cancer. Physiological Reviews. 2011;Jan1;91(1):151-175.
138. Arrieta MC et al. Alterations in intestinal permeability. Gut. 2006;55:1512:1520.
139. Alt Med Rev. Therapeutic applications of whey protein. Alternative Medicine Review. 2008;13(4):341-347.
140. Vilela RM et al. High hydrostatic pressure enhances whey protein digestibility to generate whey peptides that improve glutathione status in CFTR-deficient lung epithelial cells. Mol Nutr Food Res. 2006;50:1013-1029.
141. Kent KD et al. Effect of whey protein isolate on intracellular glutathione and oxidant-induced cell death in human prostate epithelial cells. Toxicol In Vitro. 2003;17:27-33.
142. Middleton N et al. Whole blood and mononuclear cell glutathione response to dietary whey protein supplementation in sedentary and trained male human subjects. Int J Food Sci Nutr. 2004;55(2):131-141.
资料来源:
美国梅奥诊所
www.mayoclinic.org
美国腹腔疾病基金会
http://www.celiac.org
美国腹腔疾病支持协会
http://www.csaceliacs.org
加拿大腹腔疾病协会
http://www.celiac.ca
加拿大卫生部
http://www.hc-sc.gc.ca
免责声明和安全信息
- 本信息(包括任何附带资料)不是为了取代医生或有关合格从业人士的建议或忠告。
- 任何人如果想要对本文涉及的药物、饮食、运动或其他生活方式的使用、或改变调整,以预防或治疗某一特定健康状况或疾病,应首先咨询医生或有关合格从业人士,并获得他/她们的许可。妊娠和哺乳妇女在使用本网站任何内容前,尤其应征求医生的意见。
- 除非另有说明,本网站所述内容仅适用于成人。
- 本网站所推荐的任何产品,消费者应该以实际的产品标签内容为准,尤其应关注重要的安全信息以及产品最新信息,包括剂量、使用方法和禁忌症等。
- 由于循证医学研究、文献及有关产品处于不断的变化中,本网站工作人员将尽力更新。
- 本网站不能保证所载文章内容、综合干预方案以及相关成分或产品述及的健康益处,也不承担任何责任。

